It’s a fascinating time to be in the biotherapeutics industry. What will the remarkedly rapid COVID-19 vaccine development effort teach us...
Read moreProcess Development & Validation
Process Validation for Expedited Approval Drugs
WHITE PAPER Process Validation and Regulatory Review in the Age of Expedited Approval Drugs This paper explores those dynamics and offers some revised approach suggestions....
Process Validation & Regulatory Strategies for Fast-track and Breakthrough Therapies
BioTechLogic’s Tracy TreDenick delivered a very well-received presentation at BPI West 2018 discussing process validation and regulatory strategies for expedited approval...
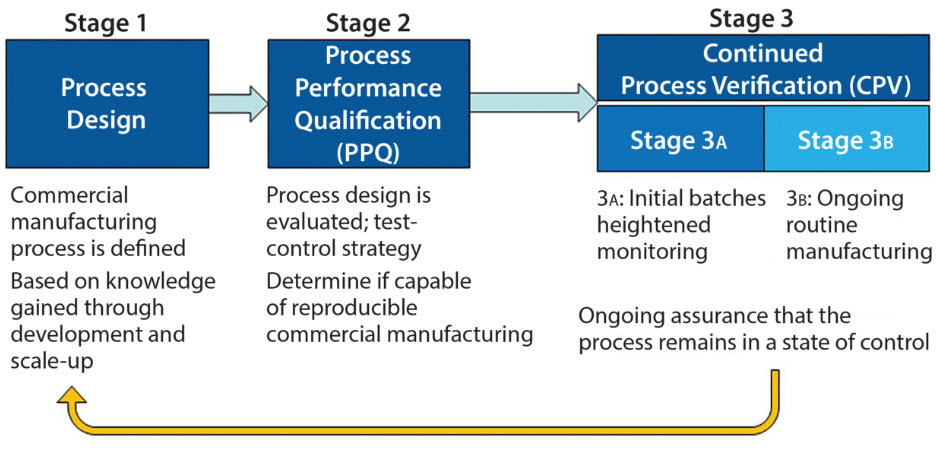
Evolution of Biopharmaceutical Control Strategy Through Continued Process Verification
As defined in the ICH Q10 guideline, a control strategy is “a planned set of controls, derived from current product and process understanding, that assures process...
A New Day for Process Validation and Regulatory Review
New drug approval and validation strategies are needed for Fast Track, Breakthrough Therapy and Accelerated Approval drugs. A new Pharmaceutical Manufacturing article,...
Brief from PDA workshop on FDA Guidance for Process Validation
BRIEF FROM THE PDA WORKSHOP ON FDA’S NEW GUIDANCE ON PROCESS VALIDATION “THE SHIFTING PARADIGM IN PROCESS VALIDATION” OCTOBER 26-27, 2009 (10/31/2009)...