Two famous pharmaceutical industry quotes encapsulate industry thinking and the push for progress in recent years. Upon...
Read moreArticles
Why Pharmaceutical Data Integrity Is More Important Than Ever
With radical pharmaceutical industry changes in the air, the importance of data integrity and the steps the pharmaceutical industry must take are clear. By Ashley Ruth,...
Live from the PDA Annual Meeting: Risk Management in Combination Products and Co-Packaged Kits
From post-aging performance testing to container closure integrity, robust design controls extend far beyond “constituent part” requirements. With the increase in complexity...
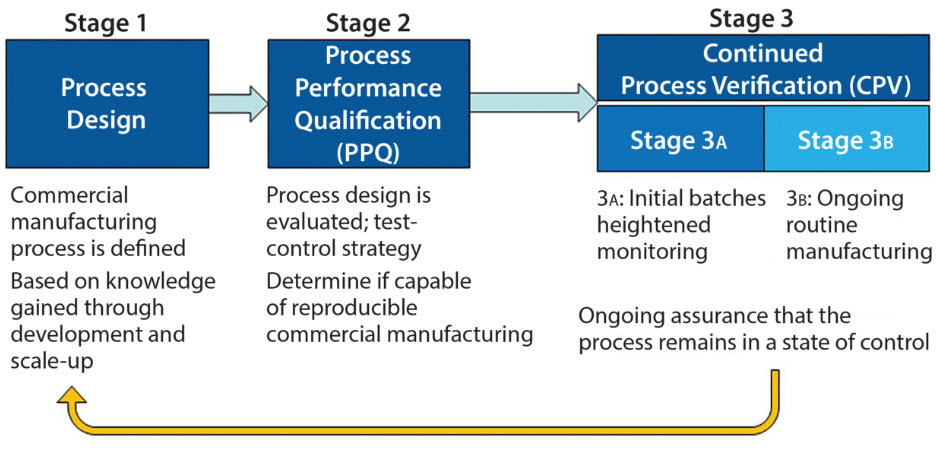
Evolution of Biopharmaceutical Control Strategy Through Continued Process Verification
As defined in the ICH Q10 guideline, a control strategy is “a planned set of controls, derived from current product and process understanding, that assures process...
Therapeutic Gene Editing: An American Society of Gene & Cell Therapy White Paper
Interested in learning more about gene editing and its therapeutic applications? Download a new white paper from the American Society of Gene & Cell Therapy (ASGCT). The...
Critical Steps for Outstanding Biopharmaceutical Project Management
Note: BioTechLogic is proud to be featured in the December 2016 issue of Pharmaceutical Manufacturing magazine. The global biopharmaceutical market is expected to grow at a...