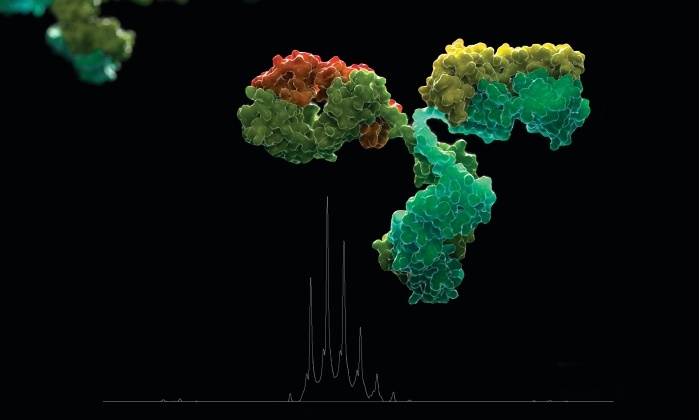
Proteins—especially antibodies—are gaining in popularity in the pharmaceutical industry, both as drugs in their own right and as targeting agents for other drugs. In fact, these so-called biologics have been part of the therapeutic landscape for so long that some have already come off patent. Last year, the U.S. Food & Drug Administration approved the first generic, or biosimilar, version of a biologic drug—Zarxio, a version of the bone marrow stimulator filgrastim made by Sandoz. And just last month, FDA approved Hospira’s Inflectra, a biosimilar version of the autoimmune disease treatment infliximab.
Protein drugs, though, aren’t as simple to characterize as the small-molecule drugs that sit in most people’s medicine cabinets. Making sure that a batch of protein drugs is highly uniform or that a generic version duplicates an original biologic is a much more complex task than it is for small-molecule therapeutics.
Click here to read the full article on Chemical & Engineering News